Floristic Investigations of Historical Parks in St. Petersburg, Russia
by Maria Ignatieva¹ and Galina Konechnaya²
¹Landscape Architecture Group, Environment, Society and Design Division, P.O. Box 84, Lincoln University, Canterbury, New Zealand
²V.L. Komarov Botanical Institute, Russian Academy of Science, 2 Professora Popova St., St Petersburg, 197376, Russia
Abstract
From 1989 to 1998, our team of researchers conducted comprehensive floristic and phytocoenological investigations in 18 historical parks in St. Petersburg, Russia. We used sample quadrats to look at plant communities; we also studied native species, nonnative species, "garden escapees," and exotic nonnaturalized woody species in numerous types of park habitat. Rare and endangered plants were mapped and photographed, and we analyzed components of the flora according to their ecological peculiarities, reaction to human influences (anthropotolerance), and origin. The entire park flora consisted of 646 species of vascular plants belonging to 307 genera and 98 families. Our analysis of species distribution in the parks showed a clear tendency toward a decrease in the number of species from the suburbs to the city center. The flora of gardens in the center of St. Petersburg was comprised mainly of weedy, meadow, and forest-meadow species and plants of open disturbed habitats. Rare herbaceous species were registered in almost all historical parks. Our study found large percentages of wetland and aquatic plants in most suburban parks, indicating that disturbances or management practices have impeded the parks' drainage systems. Our floristic investigations led us to identify ten plant indicator groups. These groups can be used for future analysis and monitoring of environmental conditions in the parks. This paper also includes analyses of plant communities in 3 of the 18 parks. Such analyses are useful for determining the success of past restoration projects in parks and other habitats and for planning and implementing future projects.
Introduction
The historical gardens and parks of St. Petersburg, Russia, are valued as monuments of landscape architecture and components of the city's urban ecosystems. They date back to the early 18th century, when Peter the Great (1672–1725) oversaw the construction of the city (his "Venice of the North") on the marshy delta of the Neva River. After World War II, intensive restoration and reconstruction was begun in almost all of St. Petersburg's historical parks. This effort has continued to this day and has employed advanced scientific methods (Ilinskaya, 1993).
The original restoration and reconstruction projects were based on detailed analyses of historic documents and on field research of plant communities (mostly inventories of canopy layers and soil maps). However, during the implementation of the projects, some mistakes were made due to misunderstandings about the ecological peculiarities of the park ecosystems. For example, light-demanding grasses were planted under the dense canopy of large deciduous trees in the Summer Garden and Tavrichesky Garden, and after only one season they began to die. In addition, heavy machinery used during construction work in many of St. Petersburg's historical parks compacted the soil and destroyed tree root systems. Consequently, there was degradation of woody plants and groundcover, including mass mortality in Tavrichesky Garden (Ignatieva, Reiman & Vorontsova, 1996) and Alexandrino Park (Subota, 1998); an intensive transition to swamp vegetation in the Nizhny (lower) Park (Rubtsova, 1996) and Alexandria Park (Ivanova & Ivanova, 1992) in Peterhof; and dying conifer species in Pavlovsky Park (Bodjurova & Karpeeva, 1995).
Because of these mistakes, and because of unfavorable environmental factors associated with modern cities in general (air and water pollution, permanent anthropogenic pressure, and harsh hydrological and climatic regimes), more restoration and reconstruction will be needed in St. Petersburg's parks. Detailed preliminary investigations of the ecology of the parks are required to avoid the mistakes of past projects—and to avoid the destruction of valuable plant communities during the design and implementation phases of future projects.
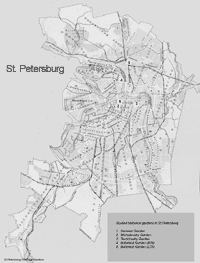
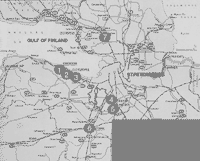
From 1989 to 1998, we and other researchers (students completing their master's thesis work under our supervision) at the St. Petersburg State Forest Technical Academy and the V.L. Komarov Botanical Institute conducted comprehensive floristic and phytocoenological investigations of the city's historic gardens (Ignatieva, 1994a, 1999; Konechnaya & Ignatieva, 1996; Rubtsova, 1996; Bodjurova & Karpeeva, 1995; Kotlyar, 1995; Volkova & Dorochova, 1994; Skosireva, 1993; Mal'kova, 1993; Starkova, 1992; Ivanova & Ivanova, 1992; Gorlanova, 1991). Eighteen of the most famous historic parks were investigated—a total research area of 2,378 hectares (5,876 acres). These were Letny Sad (the Summer Garden); Tavrichesky Garden; Mikhailovsky Garden; Shuvalovsky Park; the Verkhny (upper) and Nizhny (lower) parks in Peterhof; Alexandria Park in Peterhof; the Verkny (upper) and Nizhny (lower) parks of Oranienbaum, Ekaterininsky, and Alexandrovsky parks in Tsarskoye Selo; Pavlovsky Park; Konstantinovsky Park in Strelna; and Dvortsovy, Sylvia, Zverinets, and Prioratsky parks in Gatchina. Two other parks (those of the St. Petersburg State Forest Technical Academy and the V.L. Komarov Botanical Institute) were also included in this research because of their landscape-architectural heritage and unique botanical collections (Figures 1 and 2).
This paper compiles and analyzes the findings of these investigations. It also presents case studies examining plant communities in three of St. Petersburg's historical parks: the Summer Garden, Alexandria Park, and the White Birch region of Pavlovsky Park.
Methods
In our study of the parks, we looked at the following types of habitats: lawns, hedges, woodlands (in landscape parks), bosquets and parterres (in formal parks), flower beds, aquatic habitats (canals, ponds, and lakes), roads, and cracks in hard surfaces.
Standardized quadrats were used in all the studies to sample plant communities. For surveying the canopy layer of woody vegetation, we marked off 50-by-50-meter* quadrats and recorded the trees' composition, height, diameter, and degree of sheltering. For surveying woodland groundcover layers, we used 1-by-1-meter quadrats and recorded the general density of groundcover, along with the identity, density, height, and phenological phase of each species. For investigating meadows and lawns, we also used 1-by-1-meter quadrats.
During our investigations, voucher herbarium specimens were collected. We also mapped and photographed native and introduced ephemerals and herbs (some of them rare or endangered). Though historically less valued than the trees and shrubs in St. Petersburg's parks, these plants have both botanical and historical significance and are also very important components of park ecosystems.
Table 1: List of Higher Vascular Plants in Historical Parks and Gardens of St. Petersburg
A compiled floristic list of plants, and an analysis of this list, is shown in Table 1. The scientific names of species and families are presented according to the latest nomenclatural checklist of vascular plants of Russia and adjacent countries (Czerepanov, 1995).
Categories and Abbreviations
We organized the higher vascular plants that occurred in the parks into the following four groups according to origin (see "ORIG" column of Table 1):
- Native species (N).
- Adventive (nonnative) species that spontaneously appeared in, or were unintentionally introduced into, parks (A).
- "Garden escapees" or deliberately introduced species (G) that were planted in flower beds, lawns, and plant collections and that had naturalized in new urban habitats. These plants have different stages of naturalization.
- Exotic, nonnaturalized woody species (E).
We analyzed the flora according to the following parameters:
- Ecological group (see "ECO" column of Table 1): 1-forest, 2-forest-meadow (edge), 3-weedy-forest, 4-meadow, 5-weedy, 6-open and disturbed, and 7-aquatic.
- Anthropotolerance (see "ANTHRO" column of Table 1):
a. Urbanophil plants (UPHIL)—species that prefer human-disturbed or human-altered habitats.
b. Urbanoneutral plants (UN)—species that can grow in undisturbed natural habitats as well as in human-disturbed habitats,
c. Urbanophob plants (UPHO)—species that avoid human-altered urban habitats. - Origin of introduced plants, A, G, or E (see the "ORIG" column of Table 1): Am-North America; Sib-Siberia; Eu-Europe; FE-Far East; ES-Eurasia; SF-Siberia and Far East; and FEA-Far East and North America.
Results
Floristic Investigation
The flora in the 18 parks consisted of 646 species of vascular plants belonging to 307 genera and 98 families. This comprised 576 species of wild-growing plants (515 native, 25 nonnative, and 36 "garden escapees") and 70 species of nonnaturalized exotic woody plants. The genus richest in species was Carex (33 species). Among the exotic woody plants, North American species were the most represented (20 species), and these included Thuja occidentalis, Picea pungens, Pinus strobus, Populus balsamifera, Quercus rubra, and Ribes aureum. We found 19 European woody species (including Larix decidua, Salix alba, Salix fragilis, and Philadelphus coronarius) and 10 Siberian and Far Eastern species (including Larix sibirica, Pinus sibirica, Caragana arborescens, Berberis thunbergii, Cotoneaster lucidus, and Acer ginnala).
The number of species declined in parks from the outskirts to the center of St. Petersburg. The most species-rich parks were suburban historic parks such as Oranienbaum (400 species); Pavlovsky Park (398); Zverinets, in Gatchina (369); Nizhny (lower) Park, in Peterhof (362); Alexandrovsky Park, in Tsarskoye Selo (361); Shuvalovsky Park (341); and Konstantinovsky Park, in Strelna (340). We recorded the lowest number of species in parks at the city center: the Summer Garden (163), Tavrichesky Garden (149), and Mikhailovsky Garden (147). The flora of gardens in the center of St. Petersburg was comprised mainly of urbanophil and urbanoneutral species belonging to the weedy and meadow ecological groups.
The parks of the V.L. Komarov Botanical Institute and the St. Petersburg State Forest Technical Academy were extremely interesting from a botanical standpoint as likely sources of naturalization and dispersal for garden escapees and nonnative species. They contained the highest number of such species (35 and 16, respectively). Among the most widespread of nonnative plants were Galinsoga ciliata, Tripleurospermun perforatum (Matricaria perforata), Juncus tenuis, Gagea granulosa, and Alliaria petiolata. Detailed floristic analyses of both parks can be found in Ignatieva (1994a) and Konechnaya & Ignatieva (1996).
We recorded rare herbaceous species in almost all the suburban historical parks. Poa chaixii was recorded in Gatchina, Pavlovsk, Peterhof, and Oranienbaum parks. Luzula luzuloides was found in almost all parks except the central ones (Summer, Tavrichesky, and Mikhailovsky gardens), Shuvalovsky Park, and the park of the botanical institute. The combination of Poa chaixii, Luzula luzuloides, and Poa nemoralis could be used as an excellent groundcover model or "plant signature" (Robinson, 1993) for shady woodlands. Plant signatures could help solve the problem of creating a decorative, sustainable, shade-tolerant groundcover in old St. Petersburg parks and gardens.
German and Scandinavian botanists believe that Poa chaixii and Luzula luzuloides appeared in European parks via lawn-seed mixtures during the late 18th and early 19th centuries—a period of busy development for landscape parks in Europe (Nordhagen, 1954; Nath, 1990). Landscape parks were characterized by vast open and shaded lawns. For the shaded lawns in Germany, for example, mixtures of Poa nemoralis and Festuca rubra were widely used. But these mixtures were also contaminated with Poa chaixii and Luzula luzuloides. After several years of coexistence, all these plants formed an excellent mixture for shady park locations. European botanists are also sure that grasses such as Trisetum flavescens and Arrenatherum elatius (also found in almost all historical parks of St. Petersburg) appeared in parks accidentally through lawn mixtures. There is a theory that all these plants were brought from central and southern Europe (Nordhagen, 1954; Nath, 1990).
Some rare herbaceous species were found only in particular parks. For example, Colchicum autumnale, Phyteuma orbiculare, Valeriana dioica, and Carex paniculata were found only in Zverinets (Gatchina); Phyteuma nigrum was found only in Oranienbaum; and Phyteuma spicatum was found only in Zverinets and Oranienbaum parks. Saint Petersburg botanist A. Haare (1978) has speculated that some of the rare park species such as Primula elatior, Phyteuma spicatum, Phyteuma orbiculare, and Colchicum autumnale are natural relict species of aboriginal meadows that somehow survived within the parks.
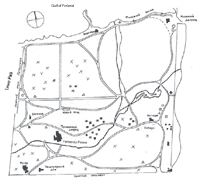
We created distribution maps for rare herbaceous species (and spring ephemerals) found in St. Petersburg's historical parks. Figure 3 shows the distribution of rare and spring species in Alexandria Park. We recommend the use of such maps for the protection of rare species during restoration.
In all suburban historical parks, the spring flora was represented by a wide spectrum of early-spring (vernal) native species such as Ficaria verna, Gagea lutea, Gagea minima, Anemonoides nemorosa, Anemonoides ranunculoides, and Corydalis solida. In the gardens at the center of St. Petersburg, profuse blooming of Gagea lutea, Gagea minima, and Facaria verna (greater than 70% groundcover) was observed only in the plant communities of the Summer Garden (Ignatieva, 1999). Hepatica nobilis was found only in Gatchina and in Pavlovsky Park, and Viola odorata and Primula elatior was found only in Dvortsovy Park and Zverinets in Gatchina. We strongly recommend protecting vernal species as high-quality groundcovers.
Our ecological and phytocoenological analyses (Ivanova & Ivanova, 1992; Rubtsova, 1996; Skosireva, 1993) of park floras showed large percentages of wetland and aquatic plants in most of St. Petersburg's suburban parks (Shuvalovsky park in St. Petersburg, and Alexandria and Nizhny parks in Peterhof, for example). The presence of these plants indicates that disturbances, such as the use of heavy machinery during construction work or poor management practices, have disrupted the parks' drainage systems. In the city-center parks, species such as Plantago major, Trifolium repens, and Poa annua were dominant, reflecting the influence of disturbances of a different kind, such as trampling, mowing, fertilizing, and construction.
As a result of our floristic investigations, we identified ten indicator plant groups. These groups can be used in future analysis and monitoring of environmental conditions in the historical parks. They reflect the ecological origin of the plants in the parks, the immigration history of the plants, and the management history of the parks.
- Nemoral plants characterizing groundcover in natural broadleaf forests: Convallaria majalis, Fragaria moschata, Anemonoides nemorosa, Anemonoides ranunculoides, Corydalis solida, Gagea lutea, and Gagea minima.
- Boreal (northern) plants characterizing typical taiga forests: Trientalis europea.
- A meadow group characterizing natural meadows: Agrostis tenuis, Anthoxanthum odoratum, Alopecurus pratensis, Alchemilla monticola, Achillea millefolium, Campanula patula, and Vicia cracca.
- Nonnative plants that arrived with lawn grass seed mixtures: Trisetum flavescens, Arrhenatherum elatius, Luzula luzuloides, Poa chaixii, Phyteuma nigrum, Phyteuma spicatum, and Pimpinella major.
- Garden escapees: Scilla siberica and Gagea granulosa.
- A group characterizing anthropogenic disturbance: Plantago major, Trifolium repens, Poa annua, Potentilla anserina, and Ranunculus repens.
- A group characterizing fertile and well-drained soils: Aegopodium podagraria, Anthriscus sylvestris, and Dactylis glomerata.
- A group characterizing wet and poorly drained soils in woodlands, edges, and lawns: Filipendula ulmaria, Lysimachia vulgaris, Calamagrostis phragmitoides, Carex vesicaria, Carex nigra, Juncus conglomeratus, Viola palustris, and Deschampsia caespitosa.
- A weedy group: Capsella bursa-pastoris, Chenopodium album, Artemisia vulgaris, and Arctium tomentosum.
- A group of aquatic plants: Glyceria maxima, Carex acuta, Potamogeton natans, and Alisma plantago-aquatica.
Vegetation Investigation
Before St. Petersburg was built, the natural landscape consisted of bogs, thickets of alder (Alnus incana) and willow (for example, Salix phylicifolia and Salix caprea), and wet conifer-deciduous forests dominated by Picea abies, Pinus sylvestris, Betula pendula, Betula pubescens, Populus tremula, and Alnus glutinosa. Peter the Great initiated an experiment to change the natural landscapes and make them into traditional European formal parks dominated by deciduous trees such as oak (Quercus), linden (Tilia), and maple (Acer). Only parts of some later-constructed (19th-century) landscape parks were based on native vegetation. These included parts of Pavlovsky Park (White Birch, Old and New Sylvia, Great Star, and Red Ponds sections), Sylvia and Zverinets parks in Gatchina, Shuvalovsky Park, Verkny (upper) Park in Oranienbaum, and Konstantinovsky Park in Strelna.
We found that the present-day plant communities for 10 of the 18 historical parks sampled were dominated by European park species (Acer platanoides, Tilia cordata, Quercus robur, Ulmus laevis, Ulmus glabra, and Fraxinus excelsior, for example). Plant associations for the 10 parks were identified as follows.
Alexandria Park in Peterhof (see case study below)
Alexandrovsky Park in Tsarskoye Selo:
- Ulmus laevis—Filipendula ulmaria—Aegopodium podagraria
- Acer platanoides—Dactylis glomerata—Aegopodium podagraria
- Quercus robur—Tilia cordata—Aegopodium podagraria—Dactylis glomerata
Dvortsovy Park in Gatchina: Quercus robur—Tilia cordata—Acer platanoides—Aegopodium podagraria—Dactylis glomerata—Filipendula ulmaria—Cirsium heterophyllum
Ekaterininsky Park in Tsarskoye Selo: Acer platanoides—Tilia cordata—Quercus robur—Aegopodium podagraria—Dactylis glomerata
Konstantinovsky Park in Strelna:
- Ulmus laevis—Filipendula ulmaria
- Acer platanoides—Aegopodium podagraria
Letny Sad/Summer Garden (see case study below)
Mikhailovsky Garden: Tilia cordata—Acer platanoides—Ulmus glabra—Poa annua—Plantago major—Taraxacum officinale
Nizhny (lower) Park in Peterhof:
- Acer platanoides—Deschampsia caespitosa
- Tilia cordata—Acer platanoides—Aegopodium podagraria
- Betula pubescens—Anthriscus sylvestris—Aegopodium podagraria
- Quercus robur—Ranunculus cassubicus—Filipendula ulmaria
- Tilia cordata—Alnus glutinosa—Equisetum pratensis
Tavrichesky Garden: Ulmus glabra—Quercus robur—Tilia cordata—Acer platanoides—Poa annua—Plantago major—Polygonum aveculare— Stellaria media
Verkhny (upper) Park in Oranienbaum:
- Tilia cordata—Luzula luzuloides
- Tilia cordata— Stellaria nemorum—Dactylis glomerata
- Tilia cordata—Calamagrostis sylvaica—Filipendula ulmaria
- Quercus robur—Tilia cordata—Calamagrostis arundinacea—Phyteuma nigra—Phyteuma spicatum—Luzula luzuloides—Trisetum flavescens
We found that all meadow plant communities in the historical parks were artificially maintained. Without regular planned management, these meadow communities would be replaced by woody pioneer plant species such as Alnus incana, Betula pendula, Salix phylicifolia, S. caprea, and Salix myrsinifolia.
We found that ephemeral plants such as Gagea lutea, Gagea minima, and Ficaria verna, along with Aegopodium podagraria and a group of weedy and meadow-forest species (Taraxacum officinale, Poa annua, and Plantago major), dominated the groundcover of the Summer Garden (Gorlanova, 1991; Ignatieva, 1994b). The mesophytic meadow grasses traditionally planted in this park, such as Poa pratensis, Festuca pratensis, and Lolium perenne, have never managed to persist due to the shady conditions there. The success of the ephemerals and Aegopodium podagraria indicates a process of stabilization of the park's ecosystem, which is very important for extending the life of the old trees and should be nurtured. Aesthetic problems with Aegopodium podagraria can be addressed using special trimming techniques prior to establishment to increase the plant's decorative qualities.
In some parks (for example, Nizhny Park and Alexandria Park in Peterhof and Dvortsovy Park in Gatchina), we found that plant communities are dominated by oak (Quercus) and other broadleaf trees in the tree layer and Filipendula ulmaria on the ground. This combination is typical of artificially created park communities in wet St. Petersburg conditions and has no analog in the native vegetation. The abundance of Filipendula ulmaria in many suburban parks indicates surplus humidity and dysfunction of the drainage system. Oak and other broadleaf trees need well-drained conditions. In time, native trees tolerant of this particular hydrological regime, such as alders and willows, will most likely replace the broadleaf trees.
Case Studies
Letny Sad: The Summer Garden (11.2 hectares; 27.6 acres)
History: In 1704, Peter the Great invited a group of talented architects and gardeners (D. Tresini, A. Schluter, I. Matveev, J.B. Leblon, I. Zhemtsov, and J. Roosen) to create a summer residence for him in the new Russian capital. Over the next 50 years, the palace, fountain system (about 50 fountains), water organ, carp pond, and amphitheatre (designed by the architect B. Rastrelli) were constructed; the formal garden was planned and planted; and 222 sculptures from Venice and Rome were installed. In 1777, a catastrophic flood destroyed many of the garden's trees, sculptures, and fountains. Since that time, the Summer Garden has been transformed into a public garden.
The Planting Design: Originally the landscape of the Summer Garden was most likely covered by spruce-birch plant communities (Picea abies—Betula pendula—Betula pubescens) on wet soils typical of the Neva River delta. The area for the garden was drained, and fertile soils were added. Initially, thousands of lime trees (Tilia cordata) and oaks (Quercus robur) from Holland and the Novgorod and Pskov regions of Russia were planted. More lime trees and oaks and other broadleaf trees such as ashes (Fraxinus), elms (Ulmus), and maples ( Acer) were planted to replace specimens killed during catastrophic floods in 1777, 1824, and 1924, and after World War II.
Current Plant Communities: In a 1989 inventory, the garden was found to contain 2,003 trees and 7,278 shrubs (Inventory of Summer Garden, 1989). The majority of trees were 50 to 100 years old. There were quite a few 150- to 200-year-old trees and 50 trees more than 200 years old. Lime trees dominated in all the plantings (more than 50% of all trees). The second most abundant were maple (Acer platanoides), followed by elm (Ulmus laevis and Ulmus glabra), oak (Quercus robur), and ash (Fraxinus excelsior). According to our floristic investigations (Gorlanova, 1991), the main type of plant community identified was Tilia cordata—Gagea minima—Gagea lutea—Ficaria verna—Aegopodium pogagraria. In some bosquets, we found small groupings of Taraxacum officinale, Ranunculus repens, and Glechoma hederacea—typical anthropogenic plants, with wide ecological ranges. Observations of the Summer Garden plant communities by Konechnaya and Ignatieva in June 2001 indicated that Aegopodium podograria was spreading successfully too. For example, many bosquets planted with typical lawn grasses (Poa pratensis, for example) in the 1990s were almost completely dominated by Aegopodium.
Gagea and Aegopodium species probably arrived in the garden as seeds in the root balls of trees that were brought from Novgorod and Pskov. These typical nemoral species found ideal conditions under the canopy of the garden's broadleaf trees. Taking into account the natural reproductive capabilities and highly competitive character of Aegopodium—a competitor species according to the Grime-Ramensky classifications (Ramensky, 1938)—as well as the absence of other natural competitors, it is not surprising that these plants have become dominant in the Summer Garden.
We found only a few turf plant communities. They were located on the slopes of the Lebyaziya Canal and the carp pond, as well as on the parterre. The turf on the slopes of the canal originated from natural meadow. Because of this, typical meadow plants such as Trifolium repens, Poa pratensis, Alopecurus pratensis, Trifolium hybridum, Galium mollugo, Alchemilla spp., Campanula rotundifolia, and Campanula glomerata (rare for a central urban park) were found there in abundance.
Flora: There were 163 species of higher vascular plants, 39 species of fungi, 14 species of mosses, and 8 species of lichens (Malisheva, Tikhomirova, Tobias, Ignatieva & Shavrina, 1995) in the Summer Garden. The nitrophylic lichens Lecanora hagenii and Scoliciosporum chlorococcum—typical indicators of air pollution—were present. However, we also found some lichens that were more characteristic of large suburban parks, such as Cetraria sepincola, Evenia prunastri, and Lecanora symmicta. These were growing in an area of the garden close to the Neva River where the higher winds most likely decreased the level of air pollution.
Alexandria Park in Peterhof (115 hectares; 284 acres)
History: Alexandria Park, an English landscape–style park, was created in the 1820s and '30s for Tsar Nicholas I. During World War II, it was almost completely destroyed. This monument of landscape architecture was virtually reborn after the war.
The Planting Design: Two-thirds of the park is located on coastal lowland between the ledge of a natural terrace and the Gulf of Finland. The remaining third is situated on the upper part of the terrace. Wetland forests of alder (Alnus) and willow (Salix) species originally covered the site. The park area was drained and 1.5 meters of fertile soil were added to the lower terrace. Thousands of oaks (Quercus robur), lime trees (Tilia cordata and Tilia platyphylos), maples (Acer platanoides), birch (Betula pendula), European mountain ash (Sorbus aucuparia), ash (Fraxinus excelsior), and also many exotic trees and shrubs (Caragana arborescens, Syringa vulgaris, Philadelphus coronarius, Cornus mas, Sambucus racemosa, Rosa majalis, Malus baccata, and Hippophae rhamnoides) were planted. Extensive meadows were grown in the front of the northern facade of the palace (the Cottage) and the Gothic Capella. Flower beds filled with exotic plants added a decorative accent.
Current Plant Communities: The dominant plant associations are Quercus robur—Filipendula ulmaria; Quercus robur—Filipendula ulmaria—Matteucia struthiopteris; and Quercus robur—Matteucia struthiopteris. There are also small areas of Quercus robur—Aegopodium podagraria and Tilia cordata—Aegopodium podagraria (Figure 4). The trees are 150 to 170 years old. All the plant communities had artificial origins; without special management and maintenance (especially drainage) they would be replaced by more moisture-tolerant natural species through succession.
The meadow is dominated by grasses (Alopecurus pratensis, Bromopsis inermis, Deschampsia caespitosa, Poa pratensis, Gliceria fluitans), legumes (Trifolium repens, Vicia cracca), Geranium palustre, Alchemilla species, Stellaria graminea, and Cirsium heterophyllum. The presence of species such as Deschampsia caespitosa, Juncus effusus, and Glyceria fluitans indicates high humidity. Most of the Alexandria meadows need permanent drainage and annual mowing to prevent the meadow plants being replaced by early successional shrubs such as Alnus incana, Salix salicifolia, and Salix myrsinifolia.
Flora: We found 317 species of higher vascular plants. Meadow plant species were the most abundant, followed by aquatic and riverside species. The high percentage of wetland species, as well as the abundance of species in the Juncaceae, suggests a process of waterlogging in the park over the last few decades.
There were six spring ephemeral and hemiephemeral native species: Anemonoides nemorosa, Anemonoides ranunculoides, Corydalis solida, Ficaria verna, Gagea lutea, and Gagea minima. Anemonoides nemorosa dominated in almost all the park's oak woodlands. The decorative ephemerals give Alexandria Park tremendous aesthetic appeal during the spring months.
We also found a number of rare species: Poa chaixii, Luzula luzuloides, Trisetum flavescens, Matteucia struthiopteris, and Melandrium dioicum (Figure 4). Poa, Luzula, and Trisetum probably appeared in Alexandria between 1830 and 1850, in the period when the main trees and lawn were planted.
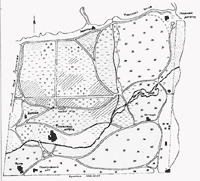
White Birch Region, Pavlovsky Park (250 hectares; 618 acres)
History: The development of Pavlovsky Park began in 1777 and continued for almost 50 years. At 600 hectares (1,483 acres), Pavlovsky is the largest of St. Petersburg's European landscape parks. Contributing to the design, based on a native forest of spruce (Picea), pine (Pinus), and birch (Betula), were Charles Cameron, who worked here from 1780 to 1786 (Slavyanka, Palace, and Great Star sections), Vincenzo Brenna, from 1796 to 1801 (Great Circles and Old and New Sylvia sections), and Pietro Gonzago, from 1801 to 1828 (White Birch, Parade Ground, and Pond Valley sections).
White Birch covers 250 hectares (618 acres) of the park. The main theme of this area is a celebration of the natural landscapes of northern Russia. There are no ponds, pavilions, or sculptures, only Gonzago's "music for the eyes," a planting style that blends native woodlands with open meadows.
The Planting Design: Gonzago worked with natural woodlands, cutting some areas to create meadows but also leaving groups of trees and single specimens to punctuate the open spaces (Figure 5). Small numbers of broadleaf trees (mainly oak, lime, and maple) were planted as well for special emphasis or accent. The original plant communities of White Birch were dominated by pine (Pinus sylvestris, 60%), spruce (Picea abies, 30%), and birch (Betula pendula, 9%). Gonzago chose birch and pine as his two major theme plants for their contrasting color, form, and texture. Oaks, limes, and maples were his planting "accompaniments."
Damage to Pavlovsky Park during World War II was catastrophic. Almost two-thirds of the trees were cut or damaged, and the drainage system was completely destroyed. All the meadows were left unmanaged and became overgrown by pioneer vegetation. During restoration after the war, a process of intensive natural regeneration of all major forest species (spruce, pine, and birch) occurred.
Current Plant Communities: Today, 65% of the trees in the White Birch region are spruce; pine only makes up 10%, and birch, 23%. Successional replacement of pine by spruce is quite evident and understandable. Picea abies plant communities are the climax type for the southern taiga zone.
The following forest associations occur here: Picea abies—Vaccinium myrtillus—Oxalis acetosella—Maianthemum bifolium (dominant type); Picea abies—Oxalis acetosella ; Picea abies—Anthyrium filix-femina—Oxalis acetosella; Picea abies—Equesetum pratense—Oxalis acetosella; Pinus sylvestris—Vaccinium myrtillus—Sphagnum spp.; Pinus sylvestris—Equisetum pratense—Oxalis acetosella; Pinus sylvestris—Athyrium filix-femina—Aegopodium podagraria; Betula pubescens—Picea abies—Vaccinium myrtillus—Oxalis acetosella; Betula pubescens —Vaccinium myrtillus; and Betula pubescens—Calamagrostis arundinacea—Oxalis acetosella.
Meadow plant communities cover the major part of the White Birch region (120 hectares; 297 acres). All meadows were replanted after World War II. The foundations of these meadows are typical grass species such as Agrostis tenuis, Anthoxanthum odoratum, Deschampsia caespitosa, Alchemilla species, Luzula multiflora, Juncus filiformis, Juncus effusus, Carex leporina, Campanula patula, Ranunculus acris, Trifolium repens, Rumex acetosa, and Lathyrus pratensis. The presence of species such Deschampsia, Luzula, and Juncus indicate naturally wet conditions and inadequate functioning of the drainage network in some places. There are a total of 82 herbaceous species and 4 woody species (Betula pubescens, Salix salicifolia, Salix aurita, and Alnus incana). The number of pioneer woody plants is quite low and directly dependent on the frequency of mowing. The average number of species per square meter varies from 11 to 20.
Flora: We found 224 higher vascular plant species in the White Birch region. Among the spring-flowering herbaceous species, Anemonoides nemorosa dominated in many forest and edge associations. We also found four protected species (Lycopodium clavatum, Platanthera bifolia, Drosera rotundifolia, and Nuphar lutea) and two rare species (Pimpinella major and Actaea spicata). Pimpinella major was probably introduced into Pavlovsky Park via seed mixtures. This particular plant dominates in the many meadow and edge communities of Pavlovsky Park.
Conclusion
Our study suggests that the most significant factor limiting floral diversity in St. Petersburg's historical parks was anthropogenic pressure, including air pollution, trampling, and disturbances such as building construction and maintenance work typical of city environments. Species diversity decreased with proximity to the city center. Large percentages of wetland and water plants in almost all the suburban parks indicated disturbance or management modification of the parks' drainage systems.
We found that ephemeral spring flora was represented by species with important ecological and decorative functions in the parks. We also found that all the suburban parks examined had a few rare species. Some of these (Poa chaixii, Luzula luzuloides, and probably Pimpinella major) could be the oldest of the parks' lawn species, while others (Primula elatior, Phyteuma spicatum, Phyteuma orbiculare, and Colchicum autumnale) could be linked to the region's ancient relict meadows. We recommend that planners of future restoration and reconstruction projects in the parks take steps to avoid or minimize damage to these species.
Through our investigations, we identified how a very old problem in the parks—creating a sustainable shade-tolerant groundcover—might be solved. We also identified ten indicator species groups for use in the monitoring of existing ecological conditions in the parks.
Our case studies of three of the St. Petersburg parks provide an example of how floristic and phytocoenological analyses can help identify historical and post-restoration pathways of succession in plant communities. This kind of data is crucial to predicting future succession in parks and other habitats so that successful restoration may be carried out and past mistakes avoided.
* Except where noted, measurements throughout this paper are in metric notation; conversions to U.S. equivalents can be obtained at http://www.onlineconversion.com/length.htm.