The Effects of Climate Change on the Vegetation of Central European Cities
by Herbert Sukopp
lnstitut für Ökologie der Technischen Universität Berlin, D-12165 Berlin, Germany
by Angelica Wurzel
Deutscher Rat für Landespflege, D-53179 Bonn, Germany
Published online February 26, 2003
Abstract
Since the 1850s the effects of global warming have been anticipated by the rise of temperature in many big cities. In addition, vegetation changes in central European cities have been well documented. This paper begins by exploring the changing urban distribution of some ruderal herbaceous species, then discusses changes in distribution and physiological changes in tree and shrub species in response to this rise in temperature. Examples of affected species covered here include Acer negundo, Ailanthus altissima, Amelanchier spicata, Berberis julianae, Buddleia davidii, Colutea arborescens, Cornus alba, C. stolonifera, Cotoneaster bullatus, Cytisus multiflorus, C. striatus, Juglans regia, Laburnum anagyroides, Ligustrum vulgare, Mahonia aquifolium, Paulownia tomentosa, Philadelphus coronarius, Platanus x hispanica, Populus x canadensis, Prunus armeniaca, P. laurocerasus, P. mahaleb, P. persica, P. serotina, Pyrus communis, Quercus cerris, Q. rubra, Q. robur, Ribes aureum, Robinia pseudacacia, Sambucus spp., Sorbus intermedia agg., Symphoricarpos albus and Syringa vulgaris. The responses of some woody scramblers and creepers are also examined. For many of these species, there was a long lag time between introduction and invasion in the wild. Phenological investigations are briefly reviewed, including studies of Aesculus hippocastanum and Tilia euchlora. Finally, the extent to which cities can act as simulators of global climate change is considered. We conclude that although other ecological and socioeconomic factors are affecting the vegetation in urban areas, many of the nonnative invasive species found colonizing cities (or naturalizing within them) originate in warmer areas and are benefiting from the more favorable climate.
Introduction: Urban Climate in Central Europe
Climatological and environmental research has led scientists to expect global warming and other climate changes to occur within the next few decades. The causes seem to be man-made. One question is what influence, if any, a warmer climate will have on flora and vegetation.
Since the 1850s the effects of global warming have been anticipated by the rise of temperature in many big cities. Cities have particular climatic characteristics (Landsberg, 1981; Kuttler, 1993):
- More air pollution: There is from 5 to 15 times more gaseous pollution; the concentration of condensation nuclei (carbon particles and other particles) is about 10 times higher than in outlying areas. Air pollution is caused by traffic, heating of buildings, power stations, and industry. Trace gases, with the exception of low-level ozone, occur at concentrations 5 to 50 times higher than usual.
- altered solar radiation: There are 5% to 15% fewer hours of sunshine, 22% to 25% less direct solar radiation, about 10% less surface albedo, and 12% more energy reflected back to the earth due to atmospheric pollution, leading to an increased net radiation of 11% at noon and 47% in the evening.
- Wind speeds are reduced by 10% to 20% due to the roughness of the city surface. There are 5% to 20% more calm days.
- The relative humidity is between 2% (winter) and 10% (summer) lower; on clear days this difference can reach 30%.
- The annual mean precipitation is up to 20% higher; however, for a variety of reasons, water generally does not percolate down through the soil but rather runs off very quickly. In other words, the vegetation in cities must contend with higher aridity than that in surrounding terrain.
The most important ecological result of these effects is higher temperature. Cities are "heat islands" or "hot spots" on the surface of the earth. How much higher the temperature is depends on the size of the city; the difference can reach 12°C on clear days, or 1°C to 2°C in yearly mean temperature.
Climatic conditions within a city can vary considerably, depending on such factors as an area's location within the city; its type of construction and paving; its density of buildings and the emanation of heat from them; and especially, its distance from large tracts of vegetation. Different climate zones, usually more or less concentric, can be distinguished within a city. A city's internal heat islands usually coincide with its built-up areas, but changes in wind direction can temporarily heat other areas as well.
The warmer climate in cities is associated with the following:
- a shorter nongrowing season (time between the first and last frosts) and less severe frosts
- a reduction in the number of frost days (to nearly half the normal amount) and snow days.
This warmer climate has the following effects on the vegetation in cities:
- a longer growing season (e.g., in Vienna, by about 10 to 20 days yearly)
- a shift in phenological phases.
Vegetation Changes in Central European Cities
Vegetation changes in central Europe have well been documented (e.g., Haeupler & Schönfelder, 1989). The interrelationships between species ranges and climate have also been investigated (Jäger, 1968). The flora and vegetation of cities have been described and mapped for a long time (Sukopp & Wittig, 1993). From this information it is possible to draw conclusions about environmental changes, including climate changes, by comparing historical data with present conditions (Sukopp, 1973).
Lists of urban plants have been published by the working group Methodik der Biotopkartierung im besiedelten Bereich (1993) and by Frank and Klotz (1990). Some examples of spontaneously growing nonnative urban plants are presented in Table 1. The cosmopolitan character of many urban plants is a tribute to the ubiquity of humanity's modification of environmental conditions and to our efficiency as agents of dispersal (Salisbury, 1961).
Many of the ruderal species in cities are short-lived plants that react quickly to climate changes. The percentage of heat-resistant plants among ruderals is high, as indicated by their origin in warmer regions. Only in favorable conditions such as those in cities are they able to build up stable populations in central Europe. The prevailing influence of climate on ruderal plants and their ability to react quickly to climatic changes make them suitable indicators. Climate changes will thus be seen in the distribution of ruderals in cities as well as in changes of their northern boundaries and their altitude tolerances in mountainous areas (Hügin, 1992, 1995).
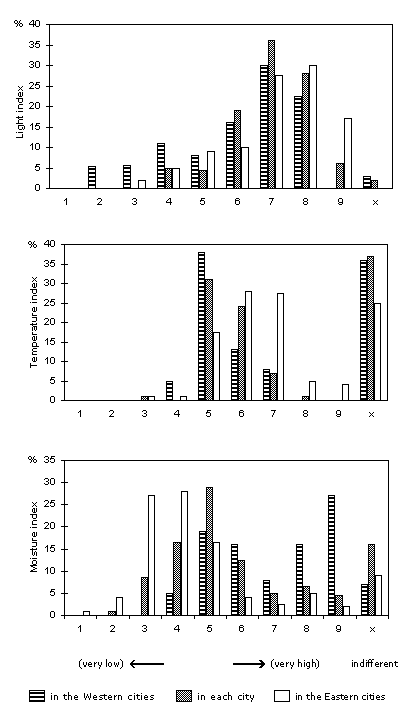
A summary of the ecological demands of the urban spontaneous flora can be attained by using indicator indices such as those of Ellenberg (1979, 1991). Kunick (1982b) used these to compare many cities with each other; the results for certain indices are shown in Figure 1. For details in Zürich, compare Landolt (2001).
Establishment of Plants Sensitive to Frost and Cold
Distribution and dispersal of nonnative plants in central Europe has changed during the past two centuries, resulting in a higher percentage of plants sensitive to frost and cold today.
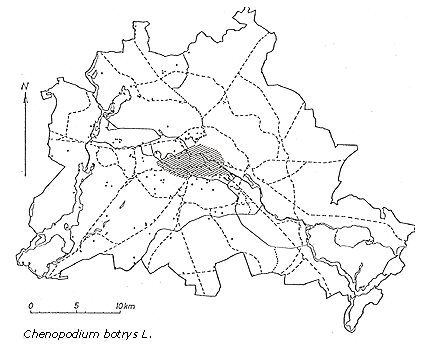
Chenopodium botrys, of south-Eurasian-Mediterranean origin, has spread over large areas of central and western Europe as a consequence of increased transportation and trade and changes in land use (Figure 2). It was introduced in Berlin in 1889 (Sukopp, 1971). The original habitats of this species are sandy and stony places along riversides and in rocky debris, very specialized habitats in which there is little competition. The plant's secondary habitats in its naturalized range in central Europe are roadsides, fields, and rubble sites. Under natural conditions there are few such sites in central Europe; only under the influence of humans has the establishment of open calcareous sites with sand and gravel increased. Chenopodium botrys has only one of the typical characteristics of weed species—high seed production under favorable environmental conditions. What is more, for germination to occur, very specialized conditions involving the quality and intensity of light, photoperiod, and storage of seeds must be met.
In central European cities, Hordeum murinum is an indicator of dry and warm sites on permeable soils. According to Wittig (1991), it has been found in cities in the northern part of central Europe since Roman times. By mapping this species at Osnabrück, Germany, in 1978 and 1989–90, Hard and Kruckemeyer (1990) showed clear expansion of its range without corresponding climate changes. They suggested that this was due to the intensification of land use and a changing concept of what constitutes a clean and well-kept city rather than climate change.
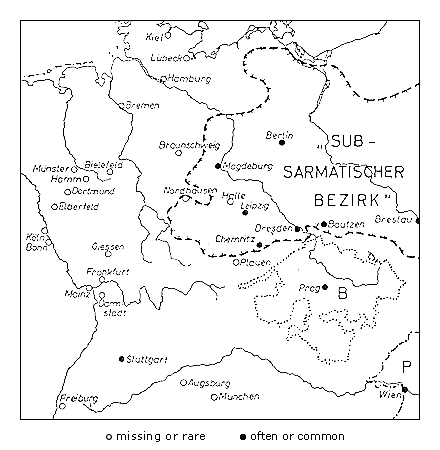
Robinia pseudoacacia has been planted in Europe since 1623 and has spread spontaneously on the rubble of bombed cities, but only in areas with relatively continental climates with warm summers (Kohler & Sukopp, 1964). In central Europe the invasion of Robinia pseudoacacia (Figure 3) has been more successful in areas with a subcontinental or sub-Mediterranean climate than in areas under oceanic influence.
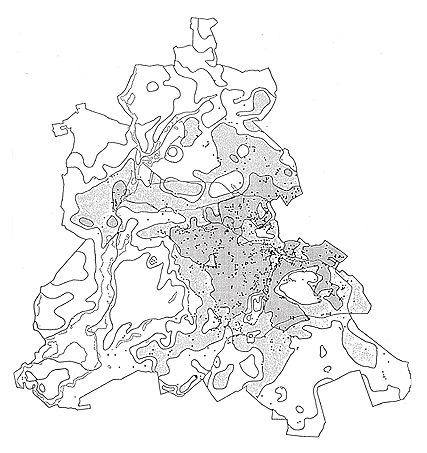
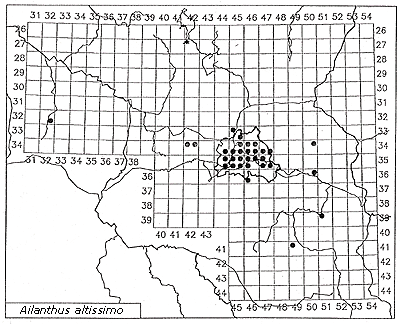
The same is true for Ailanthus altissima: In the Mediterranean or subcontinental regions, Ailanthus has spread abundantly through a broad range of sites. In central Europe, however, it is virtually confined to warmer regions or to urban-industrial sites with a more favorable microclimate. It is reasonable to assume that the spread of Robinia pseudoacacia has been promoted by the climate warming that began in the 19th century. The invasion was first observed in 1924, but only in the second part of the 19th century did this species become frequent. Similarly, the initial spread of Ailanthus was observed only at the beginning of the 20th century. Actually, in Germany it is more frequent in the warm zones of metropolitan Berlin (Figure 4, Table 3) than on the urban fringe, and in surrounding areas of Brandenburg it has been reported mainly in cities (Kowarik & Böcker, 1984; Kowarik, 1992b).
Prunus laurocerasus exemplifies how low winter temperatures may limit invasions (Adolphi, 1995). The species has been cultivated in Berlin since 1663, but the first seedlings were not observed until 1982. In regions with mild winter temperatures it obviously does better: It is common in London and in cities in the west of Germany (Table 2), and it has become a permanent member of natural forests in the sub-Mediterranean region of Italy (Gianoni et al., 1988). In Zurich, it has begun to invade forests only in recent years.
Buddleia has been invasive in Berlin since 1952, but it is actually less common there than in regions with mild winters.
Other species that are considered thermophilous, or heat-loving, with an indicator value for temperature > 7 (Ellenberg et al., 1991), include Laburnum anagyroides (with a time lag of 198 years between the plant's introduction and the onset of spreading; see also Table 3), Quercus cerris (161 years), and the sub-Mediterranean Colutea arborescens, which is native to the warmest sites in southwest Germany. In Brandenburg, 265 years elapsed before Colutea began to spread, in 1859. An even longer time lag is associated with Vitis vinifera and Juglans regia. Both have been cultivated in Brandenburg since 1200, but they were first reported as invading in 1860 and 1968, respectively. A warmer climate may also have encouraged the spread of the commonly occurring Prunus persica and of the rarer Prunus armeniaca, both of which spread 300 to 400 years after they were first cultivated (Kowarik, 1995).
An analysis of Syringa vulgaris indicates that the mode of spread also needs to be considered. This species was reported as being invasive early on, but it usually spreads by clonal growth and the enlargement of existing plantings. Even on rocky outcrops in the Rhine Valley with a favorable microclimate, fruits of Syringa do not ripen regularly (Lohmeyer & Sukopp, 1992). In Brandenburg, Syringa spreads mainly by root suckers, but recently some established shrubs were discovered fruiting along abandoned railway areas in Berlin. It is logical to assume that the fruit ripening on these sites has been promoted by the warmer urban climate. In other words, climate change has enabled the species to increase its repertoire of spreading strategies (Kowarik, 1995).
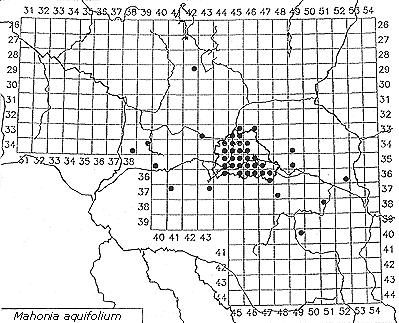
While many woody species are still in the process of establishment, one species has become a characteristic component of Robinia stands, Mahonia aquifolium. And not only does it thrive in older Robinia stands, it is also present in nearly every old Sambucus group. Ribes aureum may be another companion as well, although it cannot tolerate shade as well as Mahonia (Lohmeyer & Sukopp, 1992; Kowarik & Langer, 1994). During research in Germany's Leipzig-Bitterfeld region, Auge (1997) found several reasons why Mahonia aquifolium is such a successful invader: It can adapt easily to disturbed and even contaminated soils; it profits from clonal growth; and it is also quite fertile. See Figure 5, which shows the spontaneous distribution of Mahonia aquifolium in Brandenburg and Berlin (after Kowarik, 1992b).
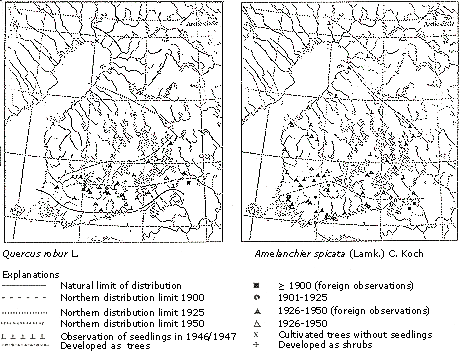
Erkamo (1956) analyzed the capability of woody species to regenerate by seeds. He found that from the beginning to the middle of the 20th century, a number of species were extending their ranges, for example Quercus robur, which is native to the south of Finland, and nonnative species such as the North American Amelanchier spicata (Figure 6).
To estimate the effect of local or global warming, it is necessary to develop models that take into account the time lags between a plant's introduction and its spread. Davis (1986) found big differences in both the magnitude and the timing of species' responses to the same climatic trend. She referred mainly to changes in abundance, demography, and distribution of native species. Different response patterns to climate changes may also be a key factor in the variation in lag times preceding biological invasions.
The examples mentioned above show that many factors have influenced the distribution of spontaneously growing flora in cities: Land use, land management, and substrate have played an important role as well as the urban climate. The plant-distribution patterns mirror building patterns and the economic structure of the city (Sukopp, 1969; Kunick, 1982a) as well as its social structure (Hard & Otto, 1985). The meso- and macro-climate of the city overlies the distribution patterns of single biotopes.
Phenological Investigations in Cities
The various temperature zones in urban areas can be illustrated by systematic phenological investigations. It is possible to create maps showing when and where a species begins a particular phase of its life cycle: budding, leafing, flowering, fruiting. The different starting points of the various phases indicate different temperature regimes in the city. It is possible to map these heat islands and cooler areas with relatively little technical difficulty.
In many cases these phenological phases start several days earlier in the center of the city than at its edge or in large parks. An analysis of the flowering phase of Tilia euchlora in West Berlin is shown in Figure 7 (Zacharias, 1972). Tilia euchlora was chosen because of the genetic homogeneity of the cultivated plants, which are offsprings of one individual. In the inner city the first flowers are seen eight days earlier than at the edge of the city. The difference in temperature between the day of flowering and one day earlier is 1°C. This accords with the distribution of phenological phases in Europe in general. In cool valleys and bogs, flowering can start two days later than at the base station for macroclimate observation. The steepest gradient is found at the boundary between forests and built-up areas.
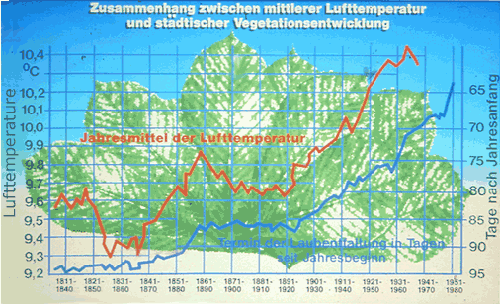
The relationship between mean air temperature and leafing of Aesculus hippocastanum in Geneva is shown in Figure 8 (Kuttler, 1987). The air temperature in Geneva has been regularly measured since 1808. From that year to the present, the date of emergence of young leaves has also been recorded (in days after the beginning of the year). Between 1808 and 1980, the date of leafing shifted from April 5 to March 5. Because this phenomenon depends on temperature, one would expect that during this time the air temperature was also changing. This proved to be the reality. After some fluctuation, the air temperature increased from 9.6°C and 9.3°C (years 1831 and 1860) to 10.4°C in 1964. Both curves show a high degree of correspondence. This example shows that plants can be indicators of climate changes in cities, provided that records have been kept long enough to make such comparisons.
Specificity of Urban Flora and Vegetation
In a study of the ruderal vegetation of Saxony, Gutte (1972) noted that urban areas are quite different biologically from surrounding areas. The urban regions of Leipzig, Chemnitz, and Dresden were differentiated on the basis of the occurrences of thermophilous plants. In his map showing the degree of occurrence of natural vegetation, Schlüter (1992) found that the flora of these big cities is unique too.
Urban flora is remarkably different from the flora of the rural hinterland. Yet a comparison of the flora of big urban agglomerations in central Europe shows little differentiation among them. The differences that do exist are caused solely by large-scale climate variations, and they increase with the distance between the urban areas. Still, the differences are astonishingly minor, even between cities as distant as Warsaw and Brussels (Kunick, 1982b).
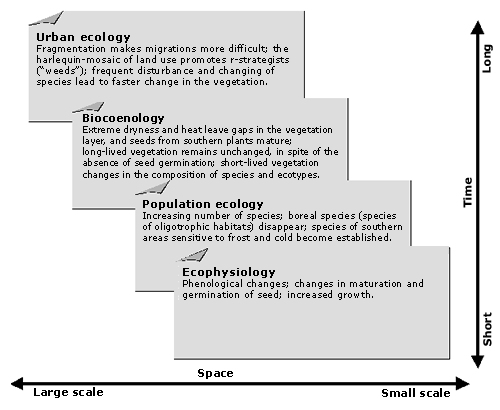
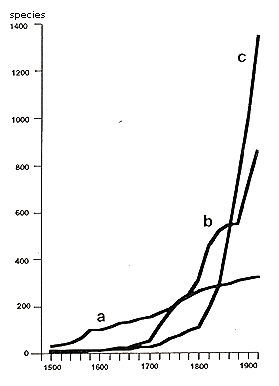
The vegetation of disturbed places—rubble fields, railway and port areas, ruins, walls, and waste areas—is "urban" in a narrow sense. The occurrence of new nonnatives on sites under human influence (Thellung, 1915) inspired many early investigations of adventive flora. The newcomers were recorded and categorized according to the time of introduction, the way they were introduced, and their degree of naturalization. Figure 9 shows the close correlation between human population growth and the number of nonnative plants. Of particular interest are the studies of the rapid colonization of bombed-out areas of London and Stuttgart, and of the climate changes of the last decade (Lousley, 1944; Salisbury, 1943). Changes associated with the urbanization of flora and vegetation, measured by time and scale, are shown in Figure 10.
Conclusions
The warmer climate is not the only factor leading to changes in the flora and vegetation of urban areas. Other ecological and socioeconomic factors also affect the vegetation.
The introduction of nonnative species via transportation and trade or for horticulture is a prerequisite for the dispersal of thermophilous plants. In many cases, these plants have crossed formerly insurmountable barriers. In cities, native plants now grow alongside those that would never have reached the new area without human help. A high percentage of nonnative species is a characteristic feature of urban floras.
Erkamo (1956) demonstrated that the rise in temperature during the first half of the 20th century caused changes in the occurrence of single plant species as well as at the community level. Over the last decade it has become evident that ecological theories on changes at the population or community level are inadequate when the climate is considered to be constant. Climate changes over time, and such change may affect plant communities, even over periods as short as decades or a few centuries (Davis, 1986).
Urban areas of central Europe are experiencing a retreat of native species and archaeophytes; at the same time they are—and will continue to be—centers of introduction and abundance of newcomers. The origin of these new species is primarily warmer regions of Europe, Asia, and the Americas.
Many native plants are also capable of colonizing new urban sites. These are called apophytes (Rikli, 1903/04). Kowarik (1992a) found 32% of Berlin's native plants established on urban sites. J.B. Falinski (personal communication, June 19, 1994) even estimated that all native plants of the Bialowieza Forest in Poland could also live as apophytes.
Plant species found in urban environments disperse relatively slowly into surrounding areas. This process can take several decades to centuries (Davis, 1986, Kowarik, 1992b). The introduced plants have quite uneven chances for dispersal.
Ten percent of all introduced species in central Europe are able to spread; 2% become permanent members of the flora; and only 1% is able to survive in natural vegetation. Robinia pseudoacacia, Acer negundo, and Ailanthus altissima were introduced in the 18th and 19th centuries. It took 100 to 180 years for these adaptable and very fertile plants to spread, although suitable sites were available before the devastation of World War II (Figure 11).
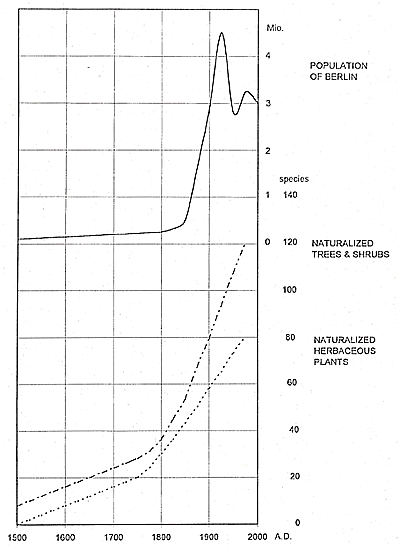
In big cities, the effects of global climate warming since the 1850s have been exacerbated by the heat-island effect. Berlin grew from a city of about 170,000 inhabitants in 1800 to a metropolitan area with 3.7 million inhabitants in 1910. Calculations of the increased warming effect of the urban climate are 0.2°C for 1798–1804, 0.7°C for 1831 to 1837, and 1.4°C for 1886 to 1898 (annual mean temperatures). For the period 1961 to 1980, there was a difference in the annual mean air temperature of more than 2°C between the center of Berlin and the surrounding areas. This warming correlates with a significant reduction of frost days: < 64 days in the center of the city; > 102 days in the surrounding areas (Stülpnagel et al., 1990). Consequently, in Berlin, an accelerated invasion of nonnative species that tolerate higher temperatures could be expected. This hypothesis is supported by Figure 12, which shows an obvious increase in woody species that began to invade Berlin by the middle of the 19th century, coinciding with the changes in temperature: In the period 1756 to 1847, the winters were colder by -0.7°C than they were in 1848 to 1907, and before 1846, extremely cold winters were much more common.
Similar trends have been observed for annual and perennial herbs in Berlin and for all established nonnative plant species in Germany. At first these trends were explained only by the huge increase in introductions and subsequent dispersal, promoted both by new transportation systems and the increasing commercial exchange of goods, which coincided with the diversification of habitats in the urban environment. But many of the nonnative invaders are native to warmer areas, and they are believed to benefit from a more favorable local climate, even on the small spatial scale of cities (Kowarik, 1995).