Elevated Ozone Levels May Lead to Strengthened Invasive Species in Urban Forests
by Eric E. Elton
Department of Environmental Sciences, University of Virginia, Clark Hall, 291 McCormick Road, PO Box 400123, Charlottesville, VA 22904-4123
Without notable advances in the protection of forests against the stresses caused by air pollution and the advance of invasive species, it is likely that dramatic diebacks and species shifts will occur in urban ecosystems (Mann, Bradley, and Hughes 1998; Fowler et al. 1999; Walther et al. 2002; Woodward, Lomas, and Kelly 2004; Carreiro and Tripler 2005; Webb, Gaston, Hannah, and Woodward 2006). Yet very few scientists have concentrated on both air pollution and nonnative species and how they combine to weaken forest ecosystems. In an effort to delve deeper into this issue, the Department of Environmental Studies at the University of Virginia has undertaken an ongoing study of the effects of ground-level ozone (O3) on mid-Atlantic urban forests. Their preliminary data suggest that some important native species (e.g., Acer rubrum, Liquidambar styraciflua, Celtis occidentalis, Quercus rubra) are less equipped to defend against oxidation reactions generated from elevated ozone levels than common invasive species (e.g., Ailanthus altissima, Morus alba, Paulownia tomentosa).
Ground-level, or tropospheric, ozone is the main constituent of industrial smog; the gas is formed by a chemical reaction of nitrous oxides (nitrogen dioxide, nitric acid, nitrates, nitrous oxide) and volatile organic compounds (benzene, formaldehyde, toluene) in the presence of sunlight.
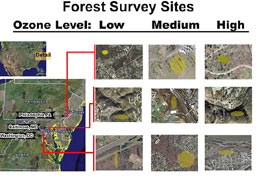
When the stomata of a plant are left open for gas exchange, ozone enters the stoma cavity and oxidizes the mesophyll cell wall, creating increased permeability of the cell wall and making the cell more vulnerable to injury. Oxidation damage in a leaf results in decreased plant performance and growth after repeated exposures. The UVA study has so far focused on leaf injuries caused by ozone damage in native and invasive trees at nine forested sites within three major East Coast cities, Washington, D.C., Baltimore, Maryland, and Philadelphia, Pennsylvania.
Within each city, UVA scientists chose three separate sites on the basis of their relative annual concentration of ground-level ozone, with thresholds set at: low (0–79 ppb), medium (80–99 ppb), and high (100–125+ ppb)1. In determining these concentration thresholds, the scientists assumed that the low levels would not injure plants, medium levels would injure only ozone-sensitive plants, and high levels would harm all but the most ozone-tolerant plants. They also assumed that high pollution levels would selectively eliminate pollution-intolerant species while augmenting establishment of pollution-tolerant species; and that forests in low-pollution areas would not have experienced this disturbance and would therefore retain native intolerant species, thus reducing the number of invasive plants that were able to establish there (Tillman 1994).
Click image to enlarge

Figure 3: Prunus serotina showing typical stipple damage due to high levels of ozone exposure. (Photo courtesy of Schaub, M., Jakob, P., Bernhard, L., Innes, J.L., Skelly, J.M., Kräuchi, N. 2002. Ozone injury database. http://www.ozone.wsl.ch. Swiss Federal Research Institute WSL, Birmensdorf.)
The study's preliminary results indicate that as ground-level ozone concentrations increase in forest settings, the native flora presence decreases, while the density of invasive species actually increases. One example of this trend shows the density of the common invasive tree of heaven (Ailanthus altissima) increase in abundance from low sites to high sites, while the native green ash (Fraxinus pennsylvanica) decreases in overall abundance. Early data also suggest that the native species studied have a greater incidence of ozone-induced oxidative injury than the invasive species. Across all sites, the plants at low-ozone-level sites experienced less overall injury than those at high concentration sites.
Native species are known to be less adaptive to changes in environmental conditions, while invasive species often cope with changes by reallocating resources and out-competing native counterparts. The ruderal nature of most invasives gives them an inherent advantage over those natives that are not able to adapt to phytotoxic gas increases.
It is clear that some plant species exhibit more tolerance to oxidative damage than others (e.g., Prunus serotina is more tolerant than Fraxinus americana) (Schaub et al. 2003), which may be due to the function of leaf chemicals (Pell et al. 1999; Massman, Musselman and Lefohn 2000). The most important chemicals in determining the injury a plant will suffer are antioxidant chemicals. Some species produce large amounts of antioxidant chemicals (e.g., ascorbate, glutathione, superoxide-dismutase, peroxidase, polyamines, carotenoids, α-tocopherol), which may decrease initial injury in active oxygen species and reduce the recovery time from injury (Massman et al. 2000; Eltayeb et al. 2006). I plan to undertake a comparison of the leaf chemicals of congeneric native and invasive plants, as well as those of invasives and natives found in the same wooded locales. Developing an understanding of how leaf chemicals are involved in oxidative injury will illuminate their role in the interplay between air pollution and invasive ecology.
The EPA is currently considering amendments to national air quality standards for ground level ozone levels in order to address the issue of vegetation damage. Dating back to 1985, European agencies like the United Nations Economics Commission for Europe established the International Co-operative Programme Forests group to prevent another major forest dieback (UNECE 1988; Ashmore and Wilson 1994; UNECE 1999; EU 2002). However, the EPA's proposed work with regard to ground-level ozone and the European resolutions addressing ozone-forest interactions fail to recognize and assess the role of invasive species, as these issues are commonly dealt with separately. The UVA study clearly suggests that increased levels of air pollution lead to increased populations of invasive species—tying the two issues together quite strongly. In order to ensure that our forests persist intact into the future, scientists and regulators must begin to tackle the hazards of air pollution and invasive species in tandem.