A Reconstruction of the Flora and Vegetation in the Central Area of Early Medieval Kyiv, Ukraine, Based on the Results of Palynological Investigations
by Lyudmila G. Bezusko, Timur V. Bezusko, and Sergei L. Mosyakin
M.G. Kholodny Institute of Botany, National Academy of Sciences of Ukraine, 2 Tereshchenkivska St., Kyiv (Kiev) 01601 Ukraine; [email protected]
Published online November 18, 2003
Abstract
This paper provides a partial reconstruction of the main features of the flora and vegetation of the central area of the city of Kiev (Kyiv in the Ukrainian-based transliteration), Ukraine, in early medieval times. The reconstruction is based on fossil spore and pollen samples. Samples for the spore-pollen analysis were selected in 1998 and 1999 during archaeological investigations on the grounds of St. Michael's Gold-Domed Cathedral and in three adjacent areas in the hilly central part of Kiev. According to archaeological data, the samples were dated to between the 10th and 12th centuries A.D. Analysis of the fossil palynoflora yielded a general list of 102 taxa of different ranks (identified by species, genus, family, or order), 72 of which were herbaceous (62.9% to 82.1% for the four sites). Analysis of the herbaceous pollen on the species level turned up a significant number of weedy flora. The data was used to supplement prior lists of weedy and cultivated plants. A comparison of our species list with diagnostic species of modern syntaxa of ruderal vegetation gives evidence that some synanthropic plant species achieved their community-forming role only during the last millennium. The data collected and analyzed in his paper provide only a fragmentary view of the natural (nonsynanthropic) vegetation that surrounded the ancient city of Kiev. However, it includes new details and paleobotanical information on the anthropic factors influencing the formation of the urban flora and vegetation of ancient Kiev.
Introduction
Understanding the interactions between humans and the natural world is best solved using methods from both the natural sciences and historiography. The reliability of paleobotanical (or archeobotanical) reconstruction is very much enhanced by combining the tools of palynology (especially paleopalynology) and paleoethnobotany, in short, by studying microfossils alongside macrofossils. Use of spore-pollen analysis to study the cultural layers of archeological sites, especially in urbanized areas, provides insight into the anthropic, or human-influenced, components of past vegetation. This is especially true when the pollen belongs to plants that are considered indicators of economic activity—namely, crop plants and weedy species associated with human presence, and especially human economic activity, in the natural or man-altered environment.
In paleobotanical terms, the ancient city of Kiev (Kyiv in the Ukrainian-based transliteration), Ukraine's capital and the so-called "Mother of Russian Cities," is a fascinating model for study. Situated along an important historical trade route, it played a leading role in the urban development of eastern Europe, until its devastation in A.D. 1240 by the Mongol and Tatar hordes of Batu Khan (a grandson of Genghis Khan). Between the 10th and 12th centuries, Kiev was among the largest and most densely populated urban areas in Europe. It is safe to assume, then, that Kiev's flora and vegetation were considerably altered by humans during these two centuries.
Material and Methods
In our study, samples for spore-pollen analysis were selected in 1998 and 1999 during archaeological investigations on the grounds of St. Michael's Gold-Domed Cathedral and in adjacent areas in the hilly central part of Kiev. In this article, we present the results of our palynological analysis of the first series of data. The samples were dated to between the 10th and 12th centuries A.D. They came from the following early medieval sites: (1) a 10th-century Slavonic burial site at modern-day Sofia Square; (2) a 10th-century Slavonic burial site on the grounds of the present-day reconstructed St. Michael's Gold-Domed Cathedral (trench No. 6); (3) a 10th-century Scandinavian burial site on the grounds of St. Michael's Gold-Domed Cathedral (chamber No. 2, the northern corner at the western side of the cathedral's gate); (4) construction fill from a building dating from the end of the 11th century through the 12th century A.D. on the grounds of St. Michael's Gold-Domed Cathedral (trench No. 15, building No. 2).
The three selected burial sites are from roughly the same era. They can be traced to the formative years of feudal ancient Rus, which included nearly all of present-day Ukraine, Belarus, and parts of northwestern Russia. This powerful medieval Slavonic state is usually called Kievan Rus in the history books, as its capital city was Kiev. In the 10th century and the first half of the 11th century, there were no permanent buildings in the immediate vicinity of the studied burial sites. The last burials to take place there were probably in the first half of the 11th century, and urban development of the area began around this time. This historical part of ancient Kiev is known as Izyaslav City or Izyaslav-Svyatopolk City. Prince Izyaslav (Isiaslav), the son of the Kievan Grand Prince Yaroslav (Iaroslav) the Wise, built St. Dmitry Monastery here in the 1060s. St. Peter's Church was built at the monastery between 1085 and 1087 by orders of Yaropolk, the elder son of Izyaslav. St. Michael's Gold-Domed Cathedral was founded in 1108 by Svyatopolk, who was then the Grand Prince of Kiev (Tolochko, 1976; Dehtiarov & Reutov, 1999; Ivakin, 2001).
In our palynological study, pollen preparation and processing followed the standard procedure proposed by V.P. Grichuk (Pokrovskaya, 1966). The samples were treated with heavy liquids (KI + CdI2) with the specific gravity of 2.0, 2.1, and 2.2 g/cm3. For pollen identification, we used the reference pollen collections of the Department of Vascular Plants at the M.G. Kholodny Institute of Botany and of the Department of Biology at the National University "Kyiv-Mohyla Academy." We also referred to pollen-identification manuals and special palynomorphological articles on pollen analysis (Erdtman, 1943, 1957, 1965; Kupriyanova, 1965; Kupriyanova & Aleshina, 1972, 1978; Boros & Jarai-Komlodi, 1975; Bobrov, Kupriyanova & Tarasevich, 1983; Grichuk & Monoszon, 1971; Monoszon, 1950, 1973, 1976, 1985; Kazartseva, 1982; Askerova, 1987; Romanova & Bezusko, 1987; Savitsky, Bezusko, Savitska & Bezusko, 1998, and others). The scientific plant names follow the recent nomenclatural checklist of vascular plants of Ukraine (Mosyakin & Fedoronchuk, 1999). For interpreting the results of our paleopalynological analysis of the medieval spore-pollen (SP) samples, we used methodological principles and data based on previous actuopalynological studies (i.e., studies of modern pollen, as opposed to studies of fossil pollen, or paleopalynology). In particular, these studies demonstrated a dependence between SP spectra in recent and subrecent surface-soil samples and the present-day vegetation patterns in the forest and forest-steppe zones of Ukraine (Arap, 1972, 1974, 1976; Bezusko, Bezusko & Yesilevsky, 1998). The similarities revealed in these studies allow us, with a reasonable degree of probability, to extrapolate fossil SP data and thereby reconstruct basic vegetation patterns in this area of medieval Kiev.
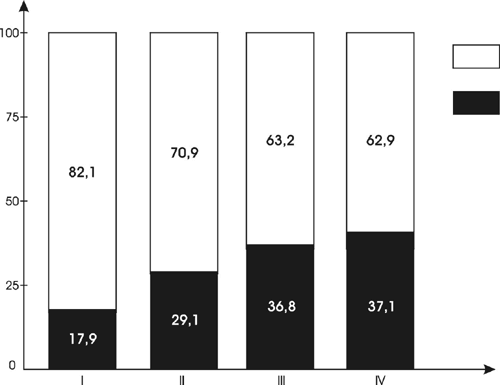
Our analysis of the fossil palynoflora yielded a general list of 102 taxa of different levels (identified by species, genus, family, or order): See Table 1. The analysis also allowed us to establish ratios between the arboreal pollen (AP, pollen of trees and shrubs) and nonarboreal pollen (NAP, pollen of mostly herbaceous plants) content of the fossil spore-pollen spectra. As shown in Figure 1, all SP spectra are characterized by some domination of pollen from herbaceous plants (62.9% to 82.1%). Of course, the ratio of pollen from nonarboreal plants to pollen from arboreal plants in the fossil SP spectra provides only generalized information indicating the presence and very approximate shares of corresponding plants. In modern (recent) SP spectra, ratios vary greatly seasonally and annually depending on climatic and atmospheric conditions, phenological rhythms of flowering periods of various plants, and other factors. However, in steppe and forest-steppe zones, the corresponding ratios in recent and subrecent samples are usually lower.
Trees and Shrubs of Ancient Kiev
The list of arboreal pollen includes 30 taxa (12 identified genera and 18 identified species). The pollen of Betula pendula, Pinus sylvestris, and Tilia cordata was present in all four SP spectra. The forests surrounding the early medieval city of Kiev from the north were represented mainly by Pinus sylvestris, Alnus glutinosa, Betula pendula, Betula pubescens, Corylus avellana, Salix species, as well as other species of trees and shrubs. These species are still typical of modern-day forests of the forest zone, which lies north of Kiev. However, the data also show that the dendroflora of the ancient town was also composed of Acer platanoides, Quercus robur, Tilia cordata, Carpinus betulus, Fraxinus excelsior, Ulmus species, Viburnum species, Rosa canina, Sambucus nigra, and some other plants. These species are mostly typical of the more open plant communities of the forest-steppe zone to the south and are also abundant today, mostly in the southern parts of Kiev. Since Kiev is situated at the border of the forest and forest-steppe zones, it is not surprising to find these trees and shrubs represented.
The linden, or lime, tree, Tilia cordata, played an important economic role in ancient Kiev. It was harvested for its timber, bast, and tar, and its blossoms made into tea. It was also considered a medicinal and even sacred plant, and the practice of preserving linden trees endures to this day: The oldest linden tree in Kiev dates back 500 years and grows near the ruins of the Tithe (Desyatynna) Church (Voinstvensky, 1986).
Pollen of the walnut tree, Juglans regia, a source of nuts and high-quality wood, was identified in the SP spectra. Historical records indicate that walnut trees were first planted in the Kiev area in the gardens of the Mezhygorsky and Vydubetsky monasteries near ancient Kiev (now within modern-day Kiev). But the date of these introductions was defined rather widely, as probably sometime between the 10th and 12th centuries A.D. (Strela, 1990). Our palynological data is the first paleobotanical confirmation of the presence of this valuable cultivated species in the Kiev dendroflora of the second half of the 10th century to the 12th century A.D.
In the SP spectrum from deposits of the end of the 11th century through the 12th century A.D., pollen grains of apple trees, Malus species, were also identified. Historical sources tell of an abundance of apple gardens in the territory of early medieval Kiev. For example, chronicles of the 11th century mention the famous apple garden of the Kyivo-Pechersky Monastery (Kiev Cave Monastery) and also the gardens near the St. Sofia Cathedral (also known as the Cathedral of the Holy Wisdom) and the St. Cyril (Kirilovsky) Monastery (Uspenskaya, Klimenko, Kuznetsov & Davydenko, 1991; Kokhno & Kurdyuk, 1994). As a rule, gardens of ancient Kievan Rus were cultivated near monasteries, convents, estates of princes and boyars (members of Rus nobility, ranking immediately below the princes), as well as near houses of other town inhabitants (Uspenskaya, Klimenko, Kuznetsov & Davydenko, 1991).
Herbaceous Plants
The general list of plants identified in the SP spectra includes 72 herbaceous taxa (26 families, 7 genera, and 39 species). Analysis of the herbaceous components on the species level turned up a significant number of weedy plants, almost half of the total number.
We have compared our palynological data with paleoethnobotanical data for Kiev from the same era obtained by G.A. Pashkevich (1991, 1998): See Table 2. Notably, we have added Beta vulgaris and Malus (probably M. domestica) to Pashkevich's list of 11th- to 12th-century cultivated plants. According to A.I. Barbarych (1962), who based his work on the available historical chronicles and records, Beta vulgaris had been known in Kievan Rus since the 10th and 11th centuries A.D. It later spread to neighboring Lithuania and Poland.
It is noteworthy that A.I. Barbarych also mentions Ribes and Rubus among the plants cultivated in Kievan Rus times. We found microfossils for these genera in the 11th- to 12th-century deposits (see Table 1). These deposits (but not those from the 10th-century sites) also contained pollen grains of Sorbus species, Rosa canina s.l., Sambucus nigra, Viburnum species, Valeriana species, Convallaria majalis, Thalictrum aquilegiifolium, and other species, some of which were probably cultivated, or at least favored and preserved, in urban areas. Of the variety of weeds that appeared during the 11th and 12th centuries, Chenopodium foliosum is a species that was widely cultivated as a leaf vegetable in early medieval times. Of course, in addition to the "standard" set of crops, many weedy species were occasionally used, in Kievan Rus and elsewhere in medieval Europe, as "famine food" or as complementary additions to the standard diet of the medieval population.
We have made additions to Pashkevich's list (1991, 1998) of weedy flora of ancient Kiev (Table 2). Among these species, newly listed archaeophytes (nonnative taxa introduced before the end of the 15th century) are represented by Centaurea cyanus, Scleranthus annuus, Spergula arvensis, Atriplex sagitata, Chenopodium foliosum, Cichorium intybus, Sonchus arvensis, Fallopia convolvulus, Echinochloa crusgalli, Setaria glauca, Setaria viridis. Newly listed apophytes (native taxa) are represented by Artemisia absintium, Tussilago farfara, Cerastium arvense, Atriplex tatarica, Chenopodium botrys, Chenopodium glaucum, Chenopodium rubrum, Polycnemum arvense, and Plantago lanceolata. New additions to the list of euapophytes (typical apophytes) are Arctium tomentosum, Artemisia scoparia, Artemisia vulgaris, Cirsium arvense, Echium vulgare, Stellaria media, Chenopodium album s.l., Taraxacum officinale s.l., Chelidonium majus, Plantago major, Plantago media, Polygonum aviculare s.l., Rumex crispus, Rumex confertus, Galium aparine, and Equisetum arvense.
From these data, we can confirm the immigration and naturalization status of many of the weedy species of present-day Kiev, as reported by V.V. Protopopova (1991). The most essential changes we've made are to the group known as the kenophytes (species that immigrated during the period from the 16th century to the present). Several weedy species should now be recognized as archaeophytes rather than kenophytes. Along with those mentioned above and others, Cannabis sativa (and its weedy races, usually known collectively as Cannabis ruderalis Janisch.) should now be considered an archaeophyte in Kiev.
The data provided here not only summarize the content of the crop and weed floras of ancient Kiev but also clearly demonstrate the prospects of further success in using paleoethnobotanical methods alongside palynological ones. The paleoethnobotanical method, for instance, considerably deepens our knowledge about grain crop species. This is because the palynological method can usually only identify the pollen of these grains to the level of "cereals" within the group collectively known in paleoethnobotanical studies as Cerealia. However, palynological materials are more informative when determining the species content of weedy flora. Undoubtedly, the most "trustworthy" taxa are those whose presence is confirmed by the two paleobotanical methods. In our study, these are the weedy species Chenopodium album s.l. and Fallopia convolvulus.
Modern-Day Descendants
We compared the species composition of the fossil weed flora (Table 2) with the diagnostic table of syntaxa of Ukraine's synanthropic vegetation compiled using the Braun-Blanquet method (Solomakha, Kostylev & Shelyag-Sosonko, 1992). The results of our analysis show that among the 36 species of weeds we reported as macrofossils and pollen remnants, 16 taxa are diagnostic species on the levels of syntaxonomic classes and orders of the modern syntaxa of ruderal and segetal vegetation of Ukraine. These syntaxa and their diagnostic species are listed below.
Class Artemisietea vulgaris Lohm., Prsg. et R. Tx. in R. Tx. 1950:
Diagnostic species: Chelidonium majus, Atriplex sagitata.
Order Artemisietalia vulgaris Lohm. in R. Tx. 1947:
Diagnostic species: Tussilago farfara.
Order Galio-Alliarietalia Oberd. ex Gors et Th. Mull. 1969:
Diagnostic species: Galium aparine.
Class Chenopodietea Br.-Bl. 1951; emend. Lohm., J. et R. Tx. 1961:
Diagnostic species: Chenopodium album, Sonchus arvensis, Fallopia convolvulus.
Order Polygono-Chenopodietalia J. Tx. et Matuszk. 1962:
Diagnostic species: Echinochloa crusgalli, Setaria viridis.
Class Galio-Urticatea Pass. 1962:
Diagnostic species: Galium aparine, Chelidonium majus.
Order Calystegietalia sepium Tx. 1950:
Diagnostic species: Galium aparine.
Class Meliloto-Artemisietea absinthii Elias 1980:
Diagnostic species: Artemisia absinthium.
Order Meliloto-Artemisietalia absinthii Elias 1979:
Diagnostic species: Echium vulgare.
Class Plantaginetalia majoris R. Tx. et Prsg. in R. Tx. 1950:
Diagnostic species: Plantago major, Polygonum aviculare.
Class Polygono-Chenopodietea (Lohm., J. et R. Tx. 1961) Elias 1984:
Diagnostic species: Chenopodium album, Polygonum aviculare, Setaria glauca, Spergula arvensis.
Class Sisymbrio-Onopordetea (Br.-Bl. 1964) Gors 1966:
Diagnostic species: Atriplex tatarica, Chenopodium glaucum.
Our data show that 7 of the 12 syntaxa listed are represented by several different species. The simultaneous presence (in our SP spectra) of several diagnostic species for each syntaxon is indirect evidence of the existence of these or similar syntaxa in the past. Thus, we can assume that in ancient Kiev, several paleosyntaxa of ruderal vegetation were already well developed by the 10th, 11th, and 12th centuries, and these paleosyntaxa were similar in their ecological and phytosociological characteristics to the listed syntaxa of the present-day ruderal vegetation of Ukraine.
Syntaxa of the class Artemisietea vulgaris are widespread on moderately humid sandy, chernozem and clayey substrata. They very seldom occur on rubbly substrata. Communities of mesophytic perennials, which are mostly represented in the order Artemisietalia vulgaris, occur at present mostly within the forest and forest-steppe zones of Ukraine. The class Chenopodietea includes communities dominated by annual ruderal species growing mostly on mechanically disturbed substrata (Solomakha, Kostylev & Shelyag-Sosonko, 1992). Communities of nitrophilous mesophytes growing mostly on dry or slightly humid soils are united within the class Galio-Urticatea. The class Plantaginetalia majoris contains ruderal plant communities of open habitats, especially pastures and livestock grazing fields with condensed (trampled) and nitrified soils. The class Polygono-Chenopodietea unites segetal and ruderal communities on light sandy soils, and the class Sisymbrio-Onopordetea contains ruderal communities of annuals and biennials growing on loose substrata in well-illuminated localities, often quite close to human dwellings (Solomakha, Kostylev & Shelyag-Sosonko, 1992).
Undoubtedly, these seven paleosyntaxa differed somewhat from their modern analogues in Ukraine's synanthropic vegetation (for example, they did not include some recent immigrants). However, it is important to note that the modern analogues of most of these paleosyntaxa occur within the studied area at present.
Our data also provide evidence that some synanthropic plant species (in particular, many archaeophytes and all kenophytes) achieved their community-forming role in ruderal and segetal vegetation only during the last millennium, as suggested earlier (Kostylev, Bezusko, Gotun & Pashkevich, 1997). Before that, they were mostly plants of marginal and naturally disturbed habitats, and their role in well-defined plant communities was nearly negligible.
Conclusion
In conclusion, we should point out that the data collected and analyzed here provide only a fragmentary view of the natural (nonsynanthropic) vegetation that surrounded the ancient city of Kiev from the 10th to the 12th century A.D. However, this set of data includes new and valuable paleobotanical information about the anthropic factors influencing the formation of the urban flora and vegetation of early medieval Kiev. For some synanthropic plant species traditionally regarded as kenophytes (recent migrants) in the Kiev area, there is now direct paleobotanical evidence of their archaeophytic status. Botanists have often underestimated the archaeophytic content of the flora and the degree to which humans have transformed the flora. There are numerous actualistic methods (i.e., those based on the concept of actualism or uniformitarianism, and thus on modern characteristics of plants, their communities, and present ranges) for assessing the status of archaeophytes in floras (see review in Zajac, 1983-1988), but paleobotanical evidence has the last word in establishing the native-versus-introduced status and the time of immigration of synanthropic plant species.
Acknowledgments
The authors are grateful to Steven E. Clemants and Janet Marinelli (Brooklyn Botanic Garden) for their kind help and advice on the manuscript. We are also grateful to anonymous reviewers for their valuable comments.